In the seven-emirate United Arab Emirates (UAE), if you have been in other Emirates and wish to travel to Abu Dhabi, you must present at the Abu Dhabi border your negative Covid‑19 PCR test results issued within 48 hours. In essence, residents who live in Abu Dhabi but travel daily to Dubai to work must prove that they are free of Coronavirus.
Since January 2021, residents in Abu Dhabi have been queuing up at health facilities to get vaccinated against Covid-19. Likewise, Dubai offers the options of Pfizer-BioNTech and Sinopharm vaccines to citizens and residents for free starting in mid-December last year. Abu Dhabi, which has maintained a low rate (0.39%) of Coronavirus confirmed cases, does not trust Dubai.
To enjoy easy access between both Emirates, residents have no choice but to get vaccinated. While all residents and citizens are able to get the free vaccine in the UAE, people eligible to get the Pfizer-BioNTech vaccination include Emiratis and residents over the age of 60, people with chronic diseases, people of determination, frontline workers and vital sector workers.
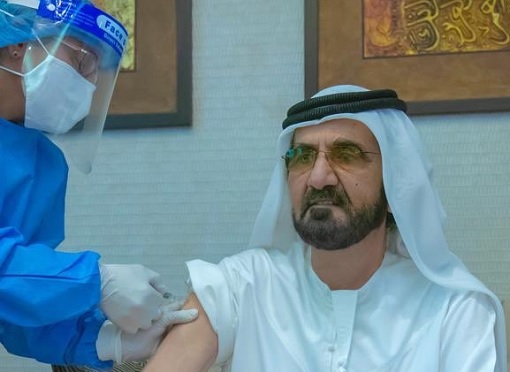
Does this mean Pfizer is the preferred vaccine and of superior quality? Not really – Dubai ruler Sheikh Mohammed bin Rashid al Maktoum in 3 Nov 2020 shared a photo on Twitter to show that he received the Sinopharm trial vaccine. The UAE only started to offer Pfizer-BioNTech vaccine in late Dec 2020, “after” the Emirate promoted the Chinese-developed Sinopharm.
In fact, the Dubai Health Authority is offering three vaccines – Sinopharm, Pfizer-BioNTech and Oxford-AstraZeneca. So, it was more about supplies and choices. While Pfizer is 95% effective in late-stage clinical trials, UAE regulators have declared that Sinopharm has 86% efficacy, and was approved for use after Phase 3 trials found it was effective against Coronavirus.
The Gulf state began Phase 3 human trials of the experimental Sinopharm vaccine as early as July, 2020. A whopping 31,000 volunteers across 125 nationalities participated in the UAE trial, a partnership between Sinopharm’s CNBG (China National Biotec Group), Abu Dhabi-based artificial intelligence company Group 42 (G42) and the Abu Dhabi Department of Health.

Unlike Pfizer-BioNTech vaccine, which is based on genetic material called mRNA, the Sinopharm vaccine is made using dead or weakened coronavirus – a common method used for decades of vaccine development including for the smallpox, flu and polio shots. But there’s one huge problem with the Chinese vaccine – lack of data and transparency.
While early Phase I and II data suggest the Chinese vaccines are safe, Chinese vaccine makers Sinopharm and the private, Beijing-based Sinovac have not released more data about their Phase III trials. The Chinese vaccine manufacturers somehow have decided that the risks of releasing more data outweigh the potential benefits. But why won’t they reveal the data?
As a start, it has been a fierce war between the U.S. and China-Russia in the competition to create Coronavirus vaccines. The Western countries will never trust the Chinese or Russian vaccines no matter what, the same way China and Russia will not buy American vaccines. But China and Russia were not interested in changing the minds of the West anyway.

The Chinese and Russian’s strategy are planting flags globally with their own homegrown vaccines – for cheap or for free. The “Vaccine Diplomacy” works like a charm because the West – the rich countries – have greedily and selfishly bought more vaccines than they need, at least 3 times more than required to vaccinate their entire populations, leaving empty shelves for the poor nations.
For example, the EU (European Union) has ordered more than 2 billion doses for its adult population of roughly 375 million, while the UK has ordered 219 million full vaccinations for its 54 million adults. Similarly, Canada has ordered 188 million full doses for its 32 million adults – enough vaccines to protect each Canadian five times, clearly a case of selfish hoarding.
In short, the rich nations, despite representing just 14% of the world’s population, have bought up 53% of the vaccines. The People’s Vaccine Alliance reveals nearly 70 low-income countries will only be able to vaccinate one in 10 people. Hence, the Chinese and Russian vaccines do not really need Phase III trials data to convince those unfortunate countries to snap up its vaccines.

Reporting the Covid-19 vaccine trial results in press releases, as in the case of China and Russia, is not enough to convince scientists that the vaccines are safe and effective. Even Western vaccine makers like Pfizer and Moderna had been criticized initially, until their Phase III trial data were publicly released in medical journals – the gold standard for gaining acceptance in the scientific community.
China’s success in flattening and defeating the pandemic, ironically, has become one of the reasons why it could not release more data. Because it does not have a good testing ground for late-stage vaccine trials in mainland, the Chinese vaccine makers have to conduct clinical Phase III trials in foreign countries such as the UAE, Indonesia, Pakistan, Turkey and Brazil.
If the Chinese wanted to cheat, they could just plug a number and claim that their vaccines are 95% effective, on par with Pfizer’s. Interestingly, Sinopharm revealed on Dec 29, 2020 that its vaccine was 79.34% effective, weeks after the United Arab Emirates said it was 86% effective. The different results suggest that China actually faces difficulty at aggregating data from multiple countries.
Likewise, Sinovac’s partners in Brazil, Indonesia, and Turkey have released four different efficacy rates for the vaccine. In Indonesia, authorities said it was 65.3% effective, but Turkish authorities said it was 91.25% effective. In Brazil, initially the authorities said it was 79% effective before saying a week later that it was only 50.4% effective.
Sinopharm and Sinovac can argue until the cows come home that the different efficacies were due to how countries have certain differences in their standards and procedures in diagnosing patients during the clinical trials. At the end of the day, the Chinese and Russian vaccine makers will still not be able to satisfy pro-Western of the discrepancies.
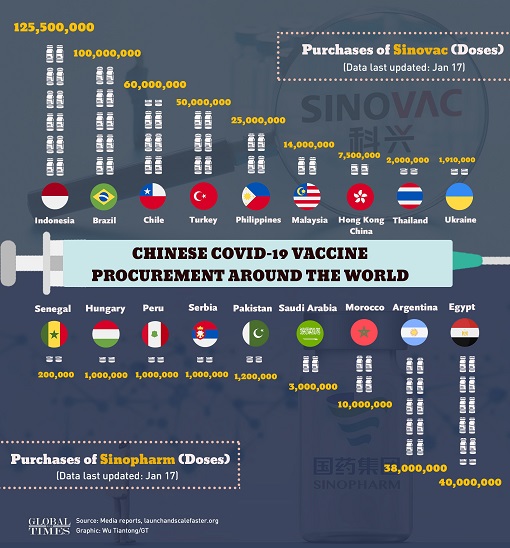
Since June 2020, the Phase III clinical trial of Sinopharm vaccine has been conducted in more than 10 countries, including the UAE, Bahrain, Egypt, Jordan, Peru, Argentina and Morocco. And 5 countries had authorized emergency use of the vaccine – China, the UAE, Bahrain, Egypt and Jordan. The UAE has already received a total of 3-million doses of the Chinese vaccine.
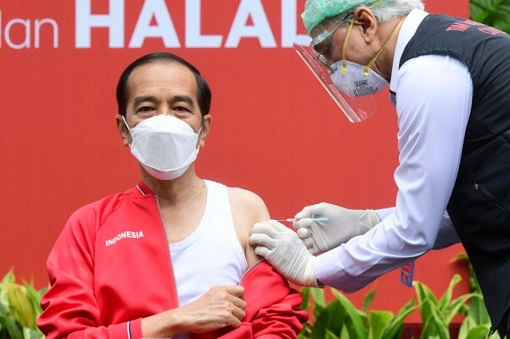
Early last month (Jan 27), Indonesian President Joko “Jokowi” Widodo received his second dose of the Covid-19 vaccine made by Sinovac Biotech at the Presidential Palace in Jakarta. The world’s fourth most populous country, Indonesia, reportedly had vaccinated 250,000 healthcare workers with the Chinese-developed Sinovac vaccine since Jokowi got his first jab.
As of this month, China is providing vaccine assistance to 53 developing countries and has export deals with 22 countries. Sinovac’s vaccine – also called CoronaVac – has gotten the green light in countries like Brazil, Turkey, Indonesia, Bolivia, Chile, Ukraine and Azerbaijan. Indonesia is one of the biggest buyers after a deal for 125 million doses of Sinovac vaccine.
Brazil is in negotiations to buy an additional 20 million doses on top of 100 million doses of Sinovac vaccine. Turkey has purchased 50 million shots of the same vaccine manufactured by Chinese drugmaker Sinovac Biotech Ltd., which can be stored in an ordinary refrigerator, making it cheaper and easier to transport than other brands that require subzero storage temperatures.

Malaysia, Singapore and the Philippines are some of the countries in Southeast Asia that have purchased Sinovac vaccine. Malaysia signed a deal in January to procure 14 million doses. Although Singapore has yet to authorise the use of Sinovac, it has already received its first shipment. Even though the Philippines have approved Sinovac, it was not for its healthcare workers.
Sinopharm’s vaccine, meanwhile, has been approved in the UAE, Bahrain, Egypt, Jordan, Iraq, Pakistan, Morocco, Peru, Serbia, Argentina and Hungary, the first European country to do so. Hungary has purchased 5 million vaccines from Sinopharm. Struggling African countries like Zimbabwe and Senegal have either received donations or made purchases of Chinese vaccines.
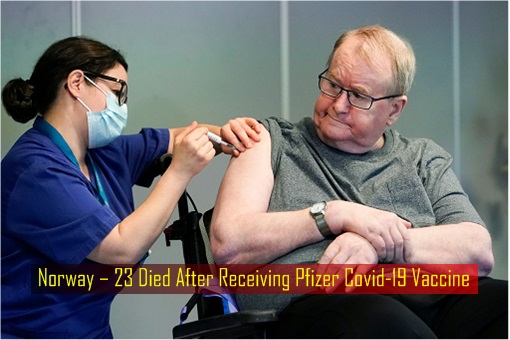
Critics may argue that an Indonesian nurse had died some 9 days after being injected with Sinovac vaccine and a Peruvian volunteer had died in Sinopharm vaccine clinical trials. But on January 14, the Norwegian Medicines Agency (Noma) reported that 23 people had died after receiving their first dose of the Pfizer-BioNTech vaccine, which was developed by the American and German pharmaceutical giants.
In Spain, 9 people died in a Spanish care home after receiving the first dose of the Pfizer-BioNTech vaccination. There were 14 deaths after Coronavirus vaccination in Belgium. In Germany, where Pfizer-BioNTech and Moderna vaccines are being used, there were 10 deaths within 4 days of Covid-19 vaccine inoculation. Obviously, Western vaccines aren’t 100% safe too.
Still, the persistent question is whether Chinese-made Coronavirus vaccines are safe. If you ask Indonesia, they would tell you that Sinovac vaccine has no critical side effects, and President Jokowi provides the best proof. However, if you read anti-Chinese news from Taiwan, critics would condemn Sinopharm vaccine as the unsafest vaccine in the world, producing 73 side effects, including loss of vision.
In the same breath, one can easily argue that despite the University of Oxford’s reputation as one of the world’s top research universities, the Oxford-AstraZeneca Covid-19 vaccine has been suspended in South Africa after “disappointing” results against a new Covid-19 variant 501.V2 or B.1.351. The vaccine, which claimed to have efficacy between 62% and 90%, depending on dosage, recorded just 21.9% efficacy in South Africa.
Early this month, the scientific journal The Lancet published new figures on CoronaVac tests that showed the vaccine was safe to use and that it induced antibodies in participants. In China, vaccines manufactured by Sinopharm and Sinovac are approved for those 59 years old and below. However, The Lancet said CoronaVac is safe and well tolerated even in older adults (60 years and older).
Perhaps the answer can be found using reverse psychology. The pandemic provides the best opportunity for Russia and China to extend their influence and prestige as scientific leaders. They are on a mission to win hearts and minds. More than 43 million doses of Sinopharm vaccine alone has been used. Do you think they would produce dubious vaccines that could kill millions of people worldwide?
If you have friends or relatives in foreign countries, don’t be surprised to hear how people in Dubai who received Sinopharm vaccine experienced zero side effects while colleagues who were given Pfizer returned home with severe fever and sore throat. Likewise, those in the U.K. experienced headache and chill after two days of Oxford-AstraZeneca vaccination.

The best practice in most countries appears to be buying all the brands available in the market – Pfizer-BioNTech, Moderna, Oxford-AstraZeneca, Sputnik V, Sinopharm and Sinovac – to ensure enough supply. To argue which vaccine is better is like comparing American iPhone and Chinese Huawei. However, people should be allowed to choose which brand they wanted and trusted the most.
Other Articles That May Interest You …
- South Africa Suspends “Ineffective” Oxford-AstraZeneca Covid-19 Vaccine – Plans To Sell It And Use J&J Instead
- Before You Rush For Covid-19 Vaccine, Remember This – You Can’t Sue Anyone If Suffer Any Serious Side Effects
- The FDA Says Pfizer Vaccine Is Safe & Effective – But There’re Many Horror Stories, Including Allergy Warning
- Forget UK’s Covid-19 Variant – This South African Coronavirus Variant Is Deadlier And Can Spread Even Faster
- Mistake Or Cheating? – AstraZeneca-Oxford Admits Manufacturing Error In Its Vaccine Effectiveness Results
- Moderna Reveals Covid Vaccine – Here’s How This 94.5% Effective Vaccine Differs From Pfizer’s Vaccine
- Coronavirus – Here’s How China Is Back To Normal While The U.S. And Europe Are Getting From Bad To Worse
- Russia Reveals “Sputnik V” – But Scientists Condemn The Coronavirus Vaccine As Risky, Dangerous & Could Backfire
- 3 Coronavirus Variants Discovered – Surprisingly, “Type-A” Found In Americans, Wuhan’s Type-B And Type-C In Europe
- The World Is Working On 20 Coronavirus Vaccines – But It Could Take Up To 18 Months
- China Appears To Be Winning The Coronavirus War, And Other Countries Are Studying How The Chinese Did It

|
|
February 27th, 2021 by financetwitter
|

|

|

|

|

|

|
Comments
Read below. Associate Editor of BMJ asks for more raw data on Pfizer as a group of “symptomatic covid-19 that were not PCR confirmed” were excluded from the effectiveness percentage.
If these numbers were added in, the speculated effectiveness percentage was just under 30%. But raw data is needed to do an accurate count.
One thing about clinical study or research done or funded by manufacturer. It will be biased towards good positive results for the manufacturer. You can do this by the multitude criterias of the study and statistics. Very few will read thoroughly and dissect the data, instead preferring to just see the conclusion paragraph with the good positive results.
Name me a study or research conducted by and/or funded by any business that shows poor bad results for them.
“You can do this by the multitude criterias of the study and statistics”.
So where can we find those, then?
Covid vaccines from China and India use the traditional dead virus method and are much safer than the experimental mRNA vaccines from MRNA and PFE. It’s a matter of time we learn about the truth.
https://childrenshealthdefense.org/defender/israel-pfizer-vaccine-killed-more-elderly-than-covid/



























It should theoretically be fine to take the risk and not get vaccinated at all.
However, this gem from me may not be good advice for a sh*t hole country like ours where Covid is going to be around like forever. That’s because we have a fcuking damn moron gomen which is designed never to get the virus under control – or to get anything right ever.
To make things worse, we have a part of the population who leaves things to AhLong and it’s up to him (his Will and Last Testament) whether you die or not you die from or get infected. Then we have another part totally fcuked up too, who are selfish or just plain fcuking stupid who would not follow instructions or even orders about taking précautions not to spread the virus.
Technically, it ain’t altogether a bad idea to get those monkeys to wipe themselves out, whoever is left of the population would hopefully bethe sane and less moronic and more useful to society.
Whatever your style of genocide, there’s also hardly any point of having any vaccine, none of them stop you from being reinfected and none of them would stop the spread of the virus.
So the moral of the story is stay panicky and kiasi. Remain confident if the gomen and our monkeys don’t cause your early and unfortunate death through Covid, they could still do their best through other countless ways, Alhamdulilah, and Ameen.